pH range of color change for indicators
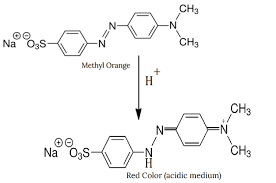
The pH range of color change for an indicator is the range of pH values over which the indicator changes color. Different indicators have different pH ranges of color change. Let me quickly remind you of something that we established in the last lesson. We saw that the pH at which the color change occurs must be the point at which pH = pKin (check the page timeline for details of the previous lesson). At this point, the color change is observed in the system and the color change usually spans through two pH units. It then follows that if we know the pKin of the indicator, we can be able to establish the pH range by counting two pH units around the value of pKin of the indicator. Let me also state that the pKin is the same as the pKa of the indicator. Let us look at phenolphthalein or instance; the pKa (pKin) is 9, the pH range is 8 – 10. For methyl orange; the pKa(pKin) is 3.5 while the pH range is 3 – 5. Finally let me say this; the pH range of color change for an indicator is de...
.png)
.jpeg)

_1.png)